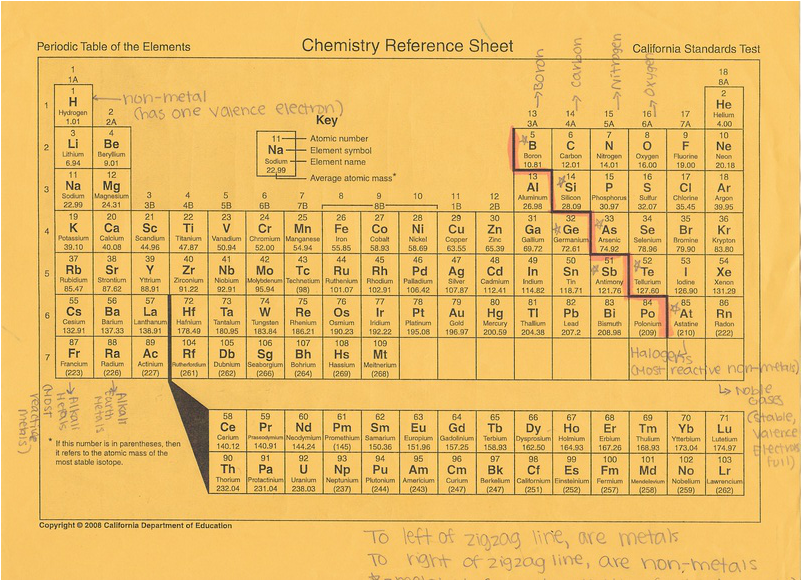
Periodic Table Notes
- Periodic Table- chart of elements organized to show similar physical or chemical properties of elements
- Energy Levels- electrons orbit the nucleus of an atom in different levels
- 7 rows- called periods; each period represents the number of occupied energy levels in each elements in the row (go across, horizontal)
- 18 columns: called groups or families; they share similar chemical and physical properties, and have the same amount of valence electrons (vertical)
Properties of metals:
- metals are good conductors of electricity and heat
- metals are shiny
- ductile (can be stretched into thin sheets)
- malleable (can be pounded into thin sheets)
- a chemical property of metal is its reaction with water which results in corrosion
- poor conductors of heat and electricity
- not ductile or malleable
- solid non-metals are brittle and break easily
- they are dull
- many non-metals are gases
- have properties of both metals and non-metals
- they are solids that can be both shiny or dull
- conduct heat and electricity better than non-metals, but not as well as metals
- ductile and malleable