Warm Ups
Date: 03.21.13
1. matter is the substance of which every physical object in the universe is made up of; matter is everything that takes up space and has mass.
2. matter is made up of atoms
3. no, you cannot see every particle within matter.
Date: 03.22.13
1. atomic number: 4, the number of protons
2. atomic mass: 9, added the protons and neutrons
3. element: beryllium, based off of the atomic number
4. protons: 4, positive charge
5. neutrons: 5, no charge
6. electrons: 4, negative charge
Date: 3/25/13
atomic number: 9
atomic mass: 19
name of the element: Fluorine
no charge because protons and electrons are equal.
Date: 03.26.13
1. matter is the substance of which every physical object in the universe is made up of; matter is everything that takes up space and has mass.
2. matter is made up of atoms
3. no, you cannot see every particle within matter.
Date: 03.22.13
1. atomic number: 4, the number of protons
2. atomic mass: 9, added the protons and neutrons
3. element: beryllium, based off of the atomic number
4. protons: 4, positive charge
5. neutrons: 5, no charge
6. electrons: 4, negative charge
Date: 3/25/13
atomic number: 9
atomic mass: 19
name of the element: Fluorine
no charge because protons and electrons are equal.
Date: 03.26.13
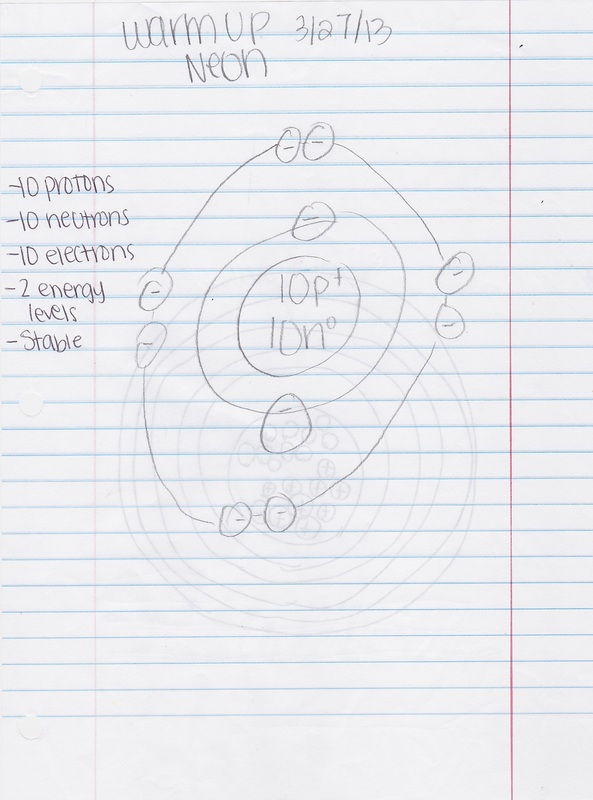
Date: 03/27/13
Date: 03.28.13
1. sodium
2. no, it is not stable
3. a positive ion
Date: 4/1/13
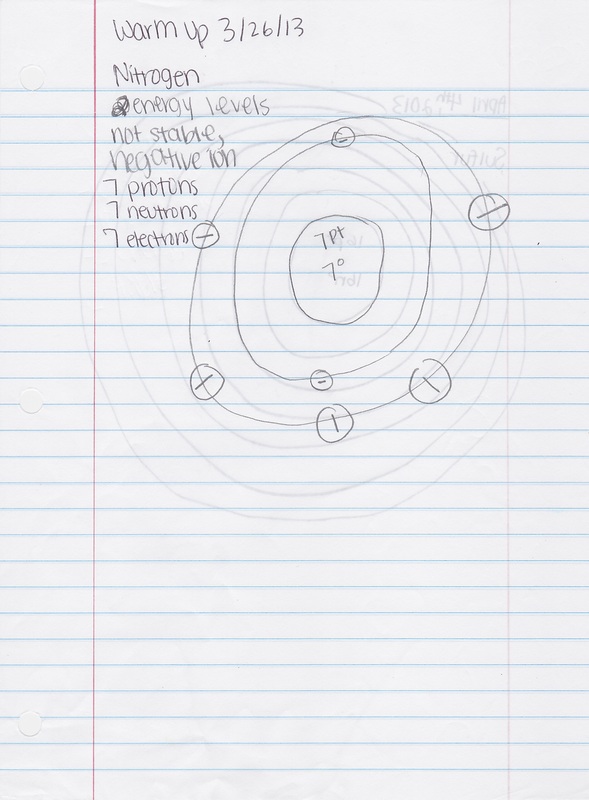
1. Nitrogen
1. sodium
2. no, it is not stable
3. a positive ion
Date: 4/1/13
1. Nitrogen
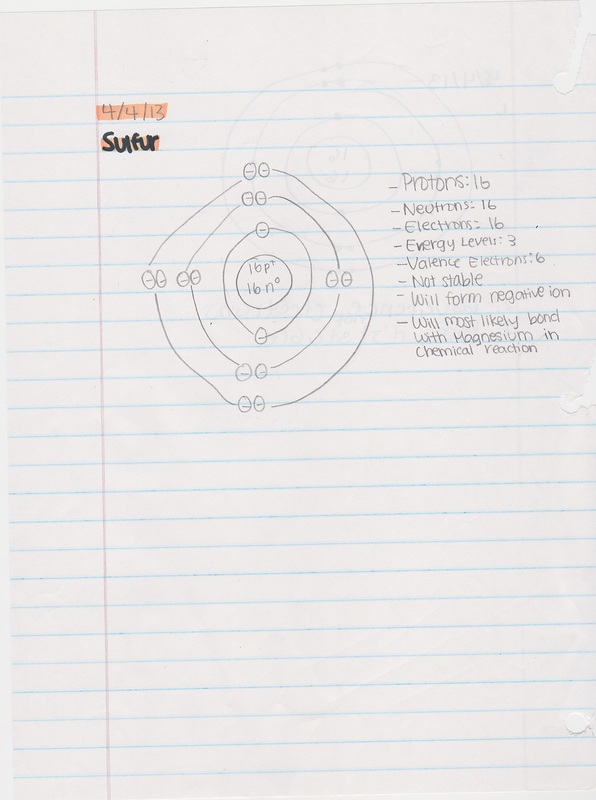
Date: 4/4/13
Date: 04.05.13
1. Magnesium
2. Carbon has 4 valence electrons, isn't stable, and would bond with Tin.
Date: 04.08.13
1. Nitrogen
2. No the valences shell is not full
3. Boron
Date: 4/10/13
1. Hg3 (PO4)2 :
3 elements and 13 atoms
2. 3P1O4:
2 elements and 15 atoms
3. Na3As1O3
3 elements and 4 atoms
Date: 4.15.13
D
Date: 04/16/13
1. A
2. 3 different elements
3. C
Date: 04.17.13
1. 0.2 Grams
2. B
Date: 04.29.13
1. Reactants: 7 atoms
Products: 6 atoms
2. 3 elements in the reactant and products side
3. No, because there are not the same amount of atoms on each side of the equation; not the same mass
Date: 05.01.13
KClO3 ---> KCl+O2
balanced equation: 2KClO3 ---> 2KCl+ 3O2
1. Magnesium
2. Carbon has 4 valence electrons, isn't stable, and would bond with Tin.
Date: 04.08.13
1. Nitrogen
2. No the valences shell is not full
3. Boron
Date: 4/10/13
1. Hg3 (PO4)2 :
3 elements and 13 atoms
2. 3P1O4:
2 elements and 15 atoms
3. Na3As1O3
3 elements and 4 atoms
Date: 4.15.13
D
Date: 04/16/13
1. A
2. 3 different elements
3. C
Date: 04.17.13
1. 0.2 Grams
2. B
Date: 04.29.13
1. Reactants: 7 atoms
Products: 6 atoms
2. 3 elements in the reactant and products side
3. No, because there are not the same amount of atoms on each side of the equation; not the same mass
Date: 05.01.13
KClO3 ---> KCl+O2
balanced equation: 2KClO3 ---> 2KCl+ 3O2